|
The purpose of corrective and preventive actions is to acquire information, analyse it and identify and investigate problems with quality in the product so ast to take appropriate and effective actions to correct the problem and prevent its recurrence. 
Basically, through this process, the company Quality Control system is able to “self-correct” and improve itself. Through a CAPA process, a company not only develops an Action Plan, but also determines, implements and monitors the effectiveness of an identified solution. Preventive action is taken to eliminate the causes of a potential deviation, non-compliance or defect in order to prevent it from occurring.Corrective action acts on existing non-compliances, deviations or defects.The Ishikawa diagram can be used as a support in brainstorming sessions with the team.ĬAPAs (Corrective Actions, Preventive Actions) are a system of quality control measures designed to eliminate the causes of a deviation, non-compliance, defect or other undesirable situation in order to prevent its recurrence. In manufacturing, the causes or factors influencing a production process are often organised into four macro-groups, which are: labour, machines (including the energy used and working and measuring instruments), materials (raw and auxiliary materials), and methods (operating procedures or practices). Ishikawa or fishbone diagram, so-called because it resembles a fishbone with a rectangle at the end containing the effect or problem.The method is usually used during problem-solving activities or in conjunction with other tools, such as the cause and effect diagram. The 5 Why method consists of asking the question “why” repeatedly until you understand all the symptoms of a problem and get to the root.It allows organisations to anticipate possible defects and errors in process design during the design phase. 
Generally (but not necessarily) the analysis is carried out beforehand and is therefore based on theoretical and not experimental considerations. Failure Mode and Effect Analysis ( FMEA) is a methodology used to analyse the failure or defect modes of a process, product or system, assess the causes and evaluate the effects on the entire system/plant.The RCA uses different models and tools such as: There are several techniques and tools that can be used to improve the approach to corrective and preventive actions. 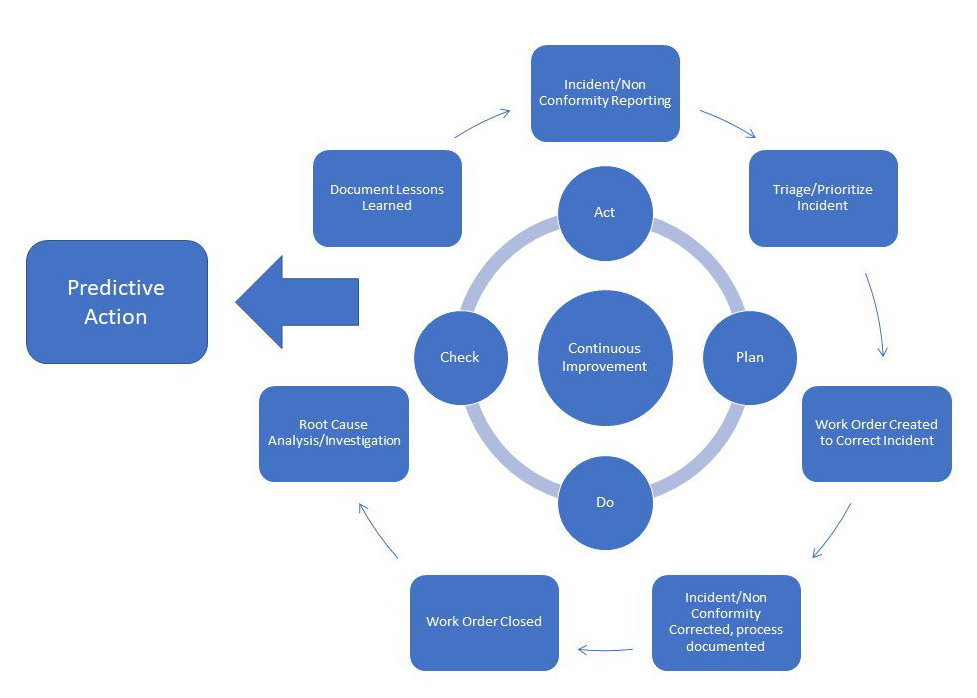
Tools and Techniques to improve the approach to preventive and corrective actions In the following paragraphs, we will reference the regulations, explore tools and techniques to improve the approach to preventive and corrective actions, answer the most frequently asked questions about CAPAs and, finally, use an infographic to demonstrate how to simplify a CAPA process using a digital system. How often is it the case that not all actions are tracked, not all steps are documented or even data is lost in a cluttered system with excessively long implementation timeframes.įurthermore, corrective action should be minimised as much as possible, as a consequence of deviations, non-conformities, etc., in favour of proactive approaches such as greater use of Quality Risk Management.īeing Proactive and Preventing is less expensive than being Reactive and Correcting! The management of CAPAs is complex and difficulties do not always depend on staff shortages, but very often they are attributable to a problem in the structure and flow of the process. Corrective and Preventive Actions (CAPA) are currently one of the major points of observation by the FDA.
1 Comment
I like that you talked about systems that can detect any possible defects that they might have as well as errors during the design phase. I can imagine how CAPA software can be a huge help in that aspect to immediately see the discrepancies and help with the improvement of the product or design before they even reach the market or your clients.
Reply
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
